Featured news
All news

How radiographer’s artwork had a brush with royalty
The artwork of two Addenbrooke’s radiographers got a royal seal of approval this week – when it was selected to appear in a display visited by King Charles and Queen Camilla.
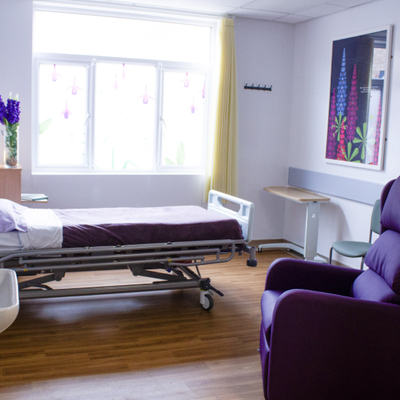
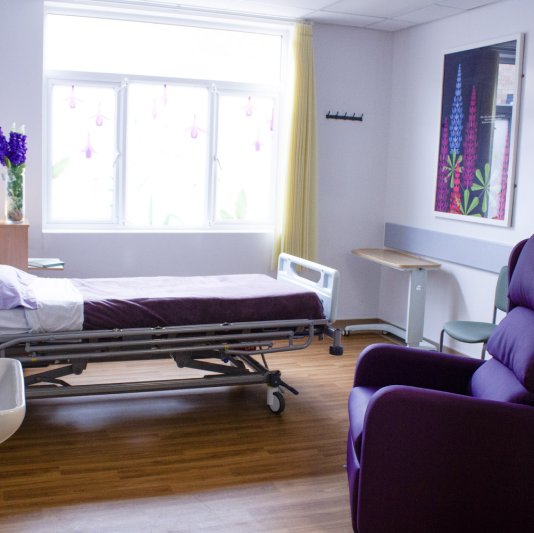
End of life care ward opens at Addenbrooke’s
A dedicated palliative and end of life care ward has opened for the first time at Addenbrooke’s, bringing together patients previously cared for across the hospital.

You Made a Difference - February 2026 - Update
Every day, across our hospital, extraordinary people go above and beyond – not for recognition, but because they care. The You Make a Difference Awards shine a light on those moments of compassion, courage and commitment that truly change lives.

Prestigious research fellowships for Trust clinicians
Dr Jyoti Nangalia, Professor James Nathan, Professor Stefano Pluchino and Professor George Vassiliou have today been named Fellows of the Academy of Medical Sciences, one of the UK’s leading scientific organisations.

You Made a Difference - April 2026
Every day, across our hospital, extraordinary people go above and beyond – not for recognition, but because they care. The You Make a Difference Awards shine a light on those moments of compassion, courage and commitment that truly change lives.

Deaf opera singer welcomes new cochlear implant trial
The national trial will include over 250 people and aims to assess the benefits of having two cochlear implants for those with profound hearing loss.

Cambridge University Hospitals announces Nicola Ayton as Chief Executive
Baroness Sally Morgan, Chair of Cambridge University Hospitals is delighted to announce the appointment of Nicola Ayton as Chief Executive.

Trust performs first robotic ‘Whipple’s procedure’ surgery
A team from Addenbrooke’s Hospital has successfully completed the most complex robotic procedure since the introduction of robot assisted surgery at Cambridge University Hospitals (CUH) more than a decade ago.

High Sheriff showcases contribution of staff and patients shaping the future Cambridge Cancer Research Hospital
The recently appointed High Sheriff of Cambridgeshire has visited the site of the new Cambridge Cancer Research Hospital yesterday (20 April) to highlight plans and discover how the project is progressing.

Cambridge scientists uncover secret of autoimmune diseases
Trust clinicians and colleagues have shown that accumulated genetic changes could contribute to autoimmunity.

You Made a Difference - March 2026
Every day, across our hospital, extraordinary people go above and beyond – not for recognition, but because they care. The You Make a Difference Awards shine a light on those moments of compassion, courage and commitment that truly change lives.

Landmark UK study could change child brain injury treatment
Researchers are calling for a re-evaluation of guidelines used in the care of children with traumatic head injuries following the first multi-centre study of its kind in the UK.

Resident doctor's industrial action - 7 April - 13 April 2026
The British Medical Association (BMA) has confirmed industrial action during April 2026.

Cambridge Children’s Hospital inaugural conference celebrates pioneering work shaping future of children’s healthcare
Over a hundred delegates joined the event in Cambridge to learn more about the new hospital’s visionary approach for holistic care, with high profile guests including the Children’s Commissioner for England Dame Rachel de Souza and broadcaster Dr Xand van Tulleken.
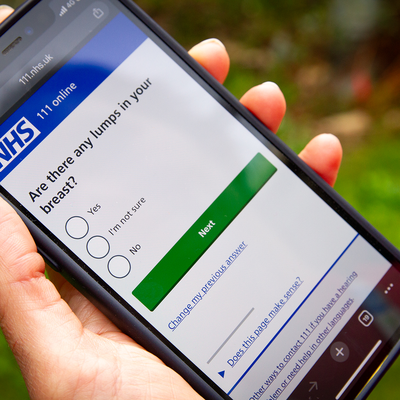
Cambs and Peterborough patients to benefit from new self-referral breast cancer service
Patients from Cambridgeshire and Peterborough with worrying breast cancer symptoms can now be referred directly to specialist clinics in hospital, without needing a GP appointment.

Trust chosen to be specialist organ perfusion centre
Addenbrooke’s has welcomed an announcement it is to become one of the first pilot assessment and recovery centres (ARC) in a bid to boost the number of life-saving organ transplants.

You Made a Difference - February 2026
Every day, across our hospital, extraordinary people go above and beyond – not for recognition, but because they care. The You Make a Difference Awards shine a light on those moments of compassion, courage and commitment that truly change lives.

Clinical Engineering expert Professor Paul White wins AHCS Honorary Fellowship
An Addenbrooke’s professor who won a prestigious award at the beginning of the year has today won another top accolade for his work.
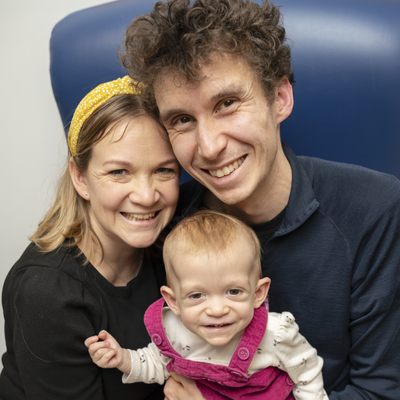
Genomic study helps detect baby’s rare growth condition
Baby Safi started treatment for a rare growth condition after participating in the Generation Study, a national genomic screening study

Global bone cement supply shortage - Update 25/02/2026
There has recently been a global shortage of medical bone cement, a material used in some orthopaedic procedures such as joint replacements. (Updated: Wednesday, 25th February 2026)