A multi-million pound trial has been announced to pave the way for the Cytosponge test to become a routine screening programme, to help detect early signs of oesophageal cancer.
The Cytosponge-TFF3 was pioneered by Cambridge University Hospitals NHS Trust (CUH) consultant gastroenterologist Professor Rebecca Fitzgerald and her team at the University of Cambridge.
Previous studies showed the test detected 10 times more cases of Barrett’s oesophagus - a condition that can lead to oesophageal cancer - compared with routine GP care.
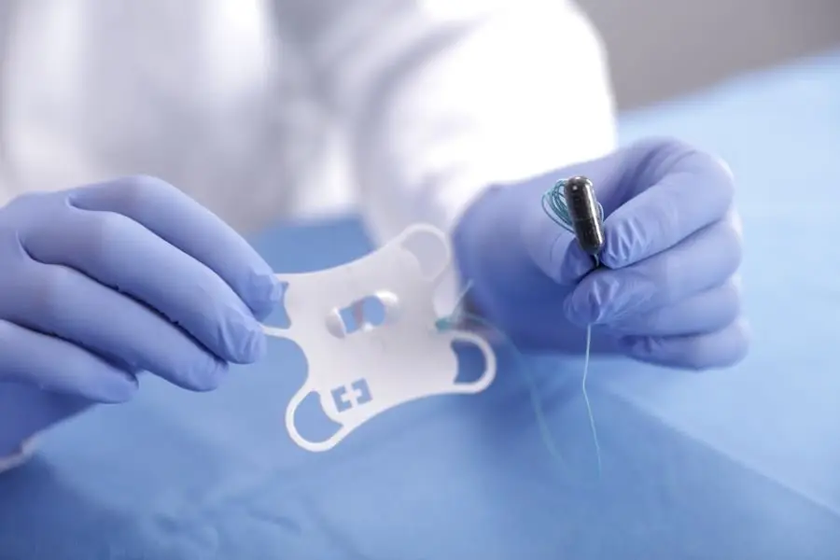
How does the Cytosponge work?
The Cytosponge begins as a pill on a string which the patient swallows.
When it reaches the stomach the coating of the pill dissolves.
This allows the pill to expand into a small sponge, about the size of a 50p coin.
The sponge is pulled back out of the stomach by a nurse.
As the sponge comes up it collects cells from the oesophagus for lab analysis.
Listen to Liz Chipchase, one of the first patients to trial the Cytosponge
Link: https://youtu.be/C52e2Qz1HOM
Cancer Research UK and the National Institute for Health and Care Research (NIHR) are providing 6.4 million pounds in funding for the new BEST4 trial.
It will explore if the Cytosponge can prevent deaths from oesophageal cancer when offered as a screening test to people on long-term medication for heartburn – one of the most common Barret’s oesophagus symptoms.
Researchers will also investigate if the Cytosponge, coupled with additional lab biomarker tests, can be used to monitor people already diagnosed with Barrett’s oesophagus instead of endoscopy, an invasive hospital procedure.
It is anticipated the trial team will begin setting up sites in autumn, with 120,000 patients to take part over 14 years.
Those involved will be randomised to different groups and a third of those will receive the Cytosponge, a quick and simple test that can be carried out by a GP.
This is an exciting opportunity to take our work on the Cytosponge-TFF3 to the next level and see whether this test not only detects more cases, but also saves lives from cancer of the oesophagus
Prof Rebecca Fitzgerald, CUH consultant
Professor Fitzgerald will lead the trial alongside Professor Peter Sasieni and his team from King’s College London.
Professor Fitzgerald said:
“The BEST4 trial will be a big piece of work, but it’s timely given the push from the NHS leadership, the Life Sciences Industrial Strategy and Cancer Research UK to detect cancers earlier in order to improve outcomes for patients.”
Dr Iain Foulkes, executive director of research and innovation for Cancer Research UK, said:
“Cancer Research UK is celebrating 120 years of life-saving discoveries this year and we’re really pleased to be funding what will hopefully be the final trial before this pioneering development is established as a screening device to detect Barrett’s oesophagus.
“There are 9,200 people diagnosed with oesophageal cancer in the UK every year and the Cytosponge will mean they can benefit from kinder treatment options if their cancer is caught at a much earlier stage, hopefully helping to boost survival rates at the same time.”

